Diagnostics: TEG, you're it
/Thromboelastography, or “TEG”, is a test that provides a visual representation of blood coagulation and clot formation in real time. While ED providers more commonly rely on PT/INR and PTT to evaluate the function of a patient’s coagulation factors, the TEG can also give us information regarding platelet function, clot strength, and fibrinolysis. And review of its core elements is crucial to maintaining facility with this dynamic test, so let’s get in for a review.
Interpretation of the TEG relies on the principle that the end result of the hemostatic process is a clot and its physical properties determine a patient’s hemostatic status. It can guide resuscitation during surgery or trauma and prevent inappropriate transfusion of blood products to non-coagulopathic patients. [1] Some have even suggested that it can be used to guide management of those who are bleeding from DOAC use.
Indications: Is it better than conventional coags?
TEG has been used as a point of care test for patients undergoing complex surgeries such as CABG or liver transplant. The test allows for earlier identification of coagulation abnormalities and more appropriate, directed transfusion resuscitation. Overall, patients whose therapy was TEG-guided required significantly fewer transfusions in the peri and post operative period; 41.5% overall transfusion incidence in the TEG group versus 65.4% in the control group. [2]
So, how does the TEG apply to us in the ED? In recent years, the test has been clinically used to guide trauma resuscitations in conjunction with conventional coagulation tests. In fact, abnormal PT/INR and PTT values have been demonstrated in systematic review to be poor predictors of bleeding prior to procedure and the ultimate need for transfusion. Ultimately, no studies reviewed found a significant risk difference between patients with normal and abnormal coags. [3] Trauma-induced coagulopathy had formerly been diagnosed using these conventional coagulation tests which, unlike the TEG, have an end point of the earliest detection of fibrin and do not assess evolution of a clot. Even when platelet count and fibrinogen are combined with these lab tests, TEG still outperforms in terms of odds ratio as a more reliable predictor of blood component transfusion, massive transfusion initiation, and mortality. In a study of almost 2000 consecutive high-level trauma activation patients at a single urban center, alpha angle less than 56 had an odds ratio alone of 8.99 in predicting massive transfusion. This was almost three times higher than any of the coagulation studies’ odds ratio. [4] These results and the overall utility of TEG in trauma have been replicated in pediatric populations as well.
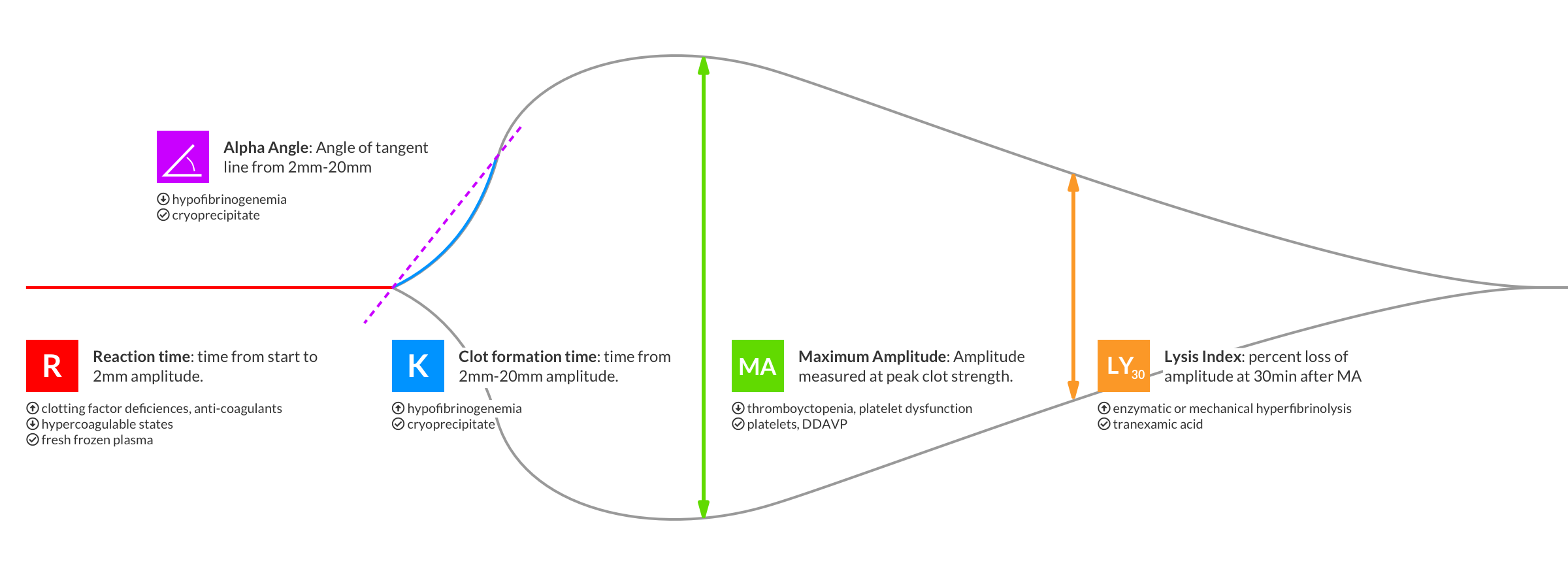
Interpretation of the TEG
Imagine you’re in trauma bay with a sick patient and as part of your trauma panel, your TEG begins to plot out on the screen behind you… how do you interpret it? To guide your resuscitation, you must first understand what each portion of the TEG means.
R-time: The reaction time, or the time it takes the coagulation cascade to generate an initial 3D network of thrombin and fibrin. It is sometimes referred to as the clot initiation time and depends on a patient’s coagulation factors. Most algorithms use prolonged R-time as a trigger for FFP transfusion.
K-time: Measures the time interval from clot initiation to a point when cross linking reaches a fixed strength, as determined by a set amplitude on the machine. In cases of severe coagulopathy, this strength may not be reached. Prolonged K time can be managed with cryoprecipitate transfusion, though some protocols do not include this measurement as it cannot be calculated in all patients.
Alpha angle: The degree angle of the tangent line from the initial TEG split point to the growing TEG curve. It represents the propagation of clot strengthening, largely mediated by fibrinogen splitting and fibrin polymerization. This measurement is used to trigger fibrinogen replacement by giving cryoprecipitate or fibrinogen itself.
MA: The maximum amplitude is the greatest strength achieved by the clot, represented by the widest part of the TEG tracing. It is most strongly correlated with not only a patient’s platelet count but more importantly, their platelet function. As we know, these two entities do not always correlate, especially in pathologic conditions. It is also important to note that in trauma patients, fibrinogen also plays a substantial role in clot strength, more so than it does in healthy patients. Still, the MA is mostly used to guide platelet transfusion therapy.
LY30: Lastly, the lysis at 30 minutes measures the percentage in amplitude reduction 30 minutes after the MA is reached. It can be thought of as the percent reduction in clot strength. All patients have some degree of normal fibrinolysis, but a provider may want to prevent excess clot breakdown in trauma. Thus, this measurement can guide us in giving TXA during a trauma resuscitation.
Limitations
The TEG unfortunately has some drawbacks. Accurate readings require multiple daily calibrations, trained lab personnel, and standardized techniques. While partial results become available within minutes, entire results take much longer. Most importantly, the TEG does not reflect the interactions between a patient’s blood and their endothelial cell surface. This dynamic is quite important in vivo.
Conclusion
Overall, the TEG has enormous utility for us during trauma resuscitations. TEG-guided resuscitation decreases the transfusion of some blood products and leads to a reduction in both hospital and ICU lengths of stay. In one study, researchers found that implementation of a TEG-guided resuscitation protocol decreased the transfusion of FFP and platelets in the first 24 hours of admission, but without any significant difference in mortality. Additionally, patients whose transfusion was TEG-guided had shorter hospital stays over conventional transfusion control groups, 11.3 days vs. 21.3 days. (5)
We are fortunate to have the means to be performing these tests and should be doing so, as conventional coagulation tests do not provide an accurate representation of the coagulopathies associated with trauma. Advancing our own personal skill sets and having the ability to visually interpret TEG plots is paramount for patient care. For practice with TEG interpretation, see this excellent post by Dr. Bernardoni.
POST BY Sarah Wolochatiuk, MD
Dr. Wolochatiuk@sarah_wolo is a PGY-1 Emergency Medicine Resident at the University of Cincinnati
PEER REVIEW BY Tim Murphy, MD AND RYAN LAFOLLETTE, MD
References
Luz, Luis Teodoro Da, et al. “Thrombelastography (TEG®): Practical Considerations on Its Clinical Use in Trauma Resuscitation.” Scandinavian Journal of Trauma, Resuscitation and Emergency Medicine, vol. 21, no. 1, 16 Apr. 2013, p. 29., doi:10.1186/1757-7241-21-29.
Shore-Lesserson, Linda, et al. “Thromboelastography-Guided Transfusion Algorithm Reduces Transfusions in Complex Cardiac Surgery.” Anesthesia & Analgesia, vol. 88, no. 2, 1999, pp. 312–319., doi:10.1097/00000539-199902000-00016.
Segal, Jodi B., and Walter H. Dzik. “Paucity of Studies to Support That Abnormal Coagulation Test Results Predict Bleeding in the Setting of Invasive Procedures: an Evidence-Based Review.” Transfusion, vol. 45, no. 9, 2005, pp. 1413–1425., doi:10.1111/j.1537-2995.2005.00546.x.
Holcomb, John B., et al. “Admission Rapid Thrombelastography Can Replace Conventional Coagulation Tests in the Emergency Department.” Annals of Surgery, vol. 256, no. 3, 2012, pp. 476–486., doi:10.1097/sla.0b013e3182658180.
Mohamed, Mohamed, et al. “The Impact of Early Thromboelastography Directed Therapy in Trauma Resuscitation.” Scandinavian Journal of Trauma, Resuscitation and Emergency Medicine, vol. 25, no. 1, 2017, doi:10.1186/s13049-017-0443-4.
Cotton, Bryan A., et al. “Rapid Thrombelastography Delivers Real-Time Results That Predict Transfusion Within 1 Hour of Admission.” The Journal of Trauma: Injury, Infection, and Critical Care, vol. 71, no. 2, 2011, pp. 407–417., doi:10.1097/ta.0b013e31821e1bf0.
Gonzalez, E., et al. “Trauma-Induced Coagulopathy: An Institution’s 35 Year Perspective on Practice and Research.” Scandinavian Journal of Surgery, vol. 103, no. 2, 2014, pp. 89–103., doi:10.1177/1457496914531927.
Gonzalez, Eduardo, et al. “Management of Trauma-Induced Coagulopathy with Thrombelastography.” Critical Care Clinics, vol. 33, no. 1, 2017, pp. 119–134., doi:10.1016/j.ccc.2016.09.002.
“Rebellion in EM 2018 - DOAC Reversal by Scott Wieters, MD.” REBEL EM - Emergency Medicine Blog, 29 Nov. 2018, rebelem.com/rebellion-in-em-2018-doac-reversal-by-scott-wieters-md/.
Schöchl, Herbert, et al. “Early and Individualized Goal-Directed Therapy for Trauma-Induced Coagulopathy.” Scandinavian Journal of Trauma, Resuscitation and Emergency Medicine, BioMed Central, 24 Feb. 2012, www.ncbi.nlm.nih.gov/pmc/articles/PMC3306198/.
Vogel, Adam M., et al. “Admission Rapid Thrombelastography Delivers Real-Time ‘Actionable’ Data in Pediatric Trauma.” Journal of Pediatric Surgery, vol. 48, no. 6, 2013, pp. 1371–1376., doi:10.1016/j.jpedsurg.2013.03.036.