Air Care Series: Ideal Resuscitation Pressure in Polytrauma with TBI
/Introduction
Traumatic brain injury (TBI), an alteration in brain function caused by an external force (1), is commonly cared for in the pre-hospital setting. TBI is responsible for a large societal burden with approximately 2.5 million TBI-related emergency department (ED) visits, 282,000 hospitalizations, and 56,000 deaths annually in the United States (2). In total, nearly 5 million patients are evaluated in emergency departments yearly in the United States alone (3). The goal of treatment in the acute pre-hospital setting focuses on the prevention of secondary injury, like that of the inpatient setting. Aggressive treatment of hypotension is critical in the prevention of secondary injury in the treatment of TBI; however, a significant amount of TBIs are associated with extra-axial trauma where permissive hypotension is often favored. In all-comers with trauma, hemorrhagic shock is the leading cause of preventable deaths (5), while more than 25% of patients with bodily trauma also have sustained a TBI (4). Appropriate pre-hospital and emergency management of patients with trauma is critical to patient survival and long-term functional outcome. The purpose of this literature review is to summarize the evidence of pre-hospital blood pressure management of patients who have sustained polytrauma and TBI, specifically for clinicians in critical care transport medicine.
Fundamentals of Cerebral Blood Flow and Autoregulation
Understanding cerebral blood flow mechanics is crucial to realizing the importance of optimal blood pressure targets following TBI. In 1783 Alexander Monro applied the principles of physics to the intracranial contents creating what came to be known as the Monro-Kellie Hypothesis (Table 1). This principle has substantial implications for cerebral blood flow during periods of intracranial pressure changes. In a healthy state, cerebral perfusion pressure remains constant due to changes in flow related to intact cerebrovascular autoregulation (CAR). CAR is often lost or impaired after severe TBI, and the intracranial pressure may be extremely sensitive to blood pressure fluctuations (7). Neuronal function becomes impaired when perfusion is 40% of normal, and irreversible damage occurs when perfusion reaches 20% of normal for a one to two hour period (8). In models of penetrating brain injury and shock, regional cerebral blood flow has been shown to decrease by more than 70% from baseline and is associated with cortical spreading depolarizations and reductions in brain tissue oxygenation (P(bt)O2) (9). Therefore, maintaining adequate cerebral perfusion pressure (CPP) is a mainstay of treatment for minimizing secondary brain injury following TBI.

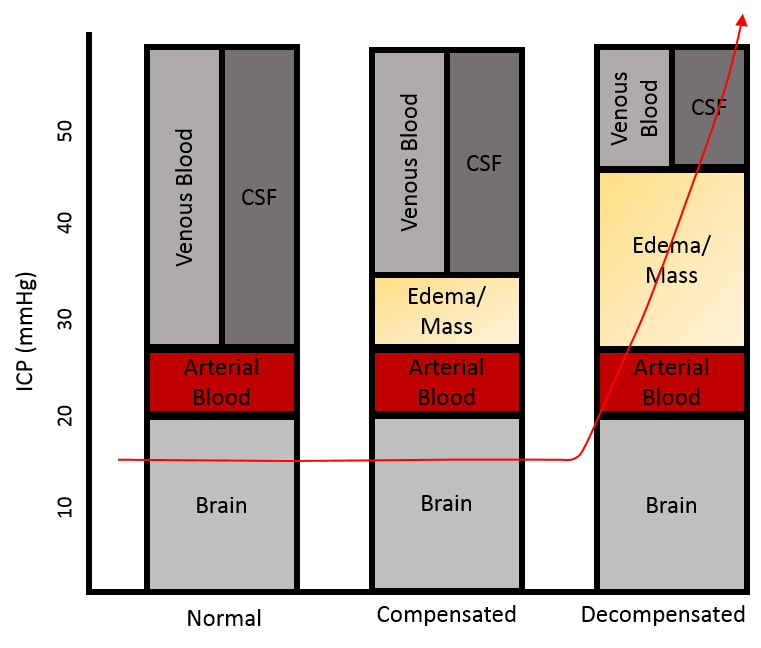
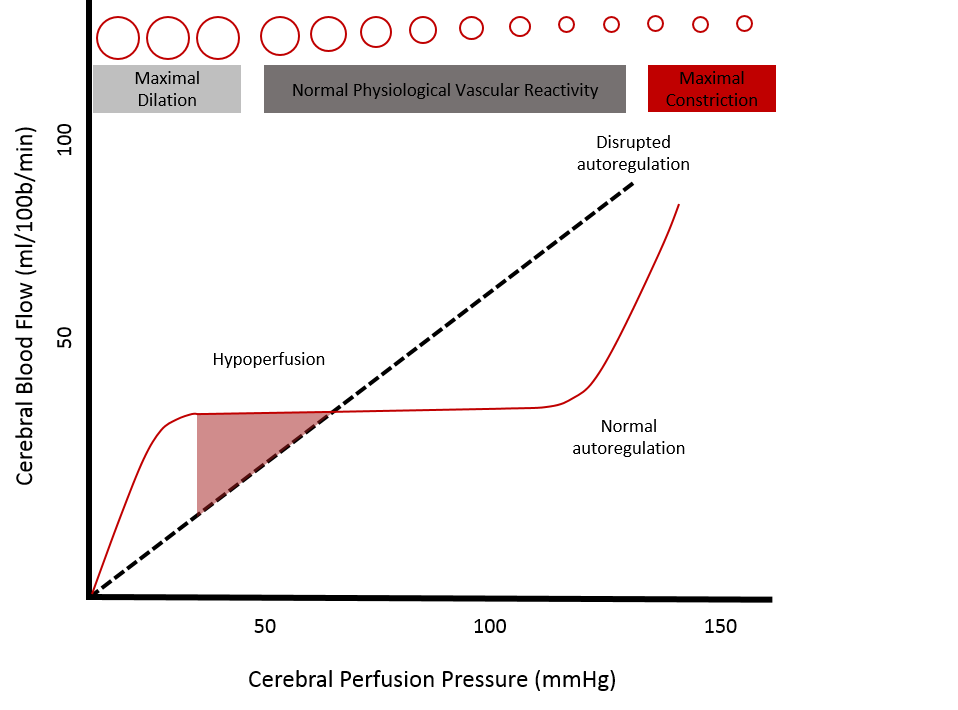
Table 1: Monro-Kellie Hypothesis ;Figure 1 (A): The Monro-Kellie Doctrine; Figure 1 (B): Cerebral Auto-regulatory Capacity
Traditional Blood Pressure Targets in Traumatic Brain Injury
Hypotension, a common sequela of trauma, is a major determinant of poor outcome in patients suffering from TBI (10, 11). Approximately 18% of patients with trauma are hypotensive on arrival to the receiving facility (12). Adequate CPP is necessary for ensuring ongoing cerebral blood flow and is calculated as the difference between mean arterial pressure (MAP) and ICP (CPP = MAP - ICP). Prevention of secondary brain injury by ensuring optimal cerebral perfusion is crucial in the pre-hospital management in patients with traumatic brain injury.
Traditionally hypotension had been defined as SBP <90mmHg. A 2009 retrospective review, which looked at patients with moderate-to-severe TBI examined thresholds of hypotension based on rates of mortality. Their recommendations were based on age, and hypotensive thresholds based on their data were SBP of 110 mm Hg for patients 15–49 years, 100 mm Hg for patients 50–69 years, and 110 mm Hg for patients ≥70 years (13), which was a change in the perceived thresholds of hypotension.
The International Mission for Prognosis and Analysis of Clinical Trials in TBI or IMPACT project was initiated in 2003 with the goal of advancing knowledge of prognosis, trial design, and treatment in TBI. In their analysis of patients who have sustained a TBI, there was a continuous relationship between systolic blood pressure and mean arterial pressure. In 8,172 patients, there was a direct linear correlation between poor Glascow Outcome Score Extended (GOSE, Table 2) and both low and high extremes in systolic blood pressure, with an optimal systolic blood pressure target around 130-140mmHg (15) (Figure 2).
A small 2012 prospective cohort study of 60 patients with isolated TBI and need for intracranial pressure monitoring assessed the relationship between systolic blood pressure (SBP), mortality rate, and recovery at 12 months. This study found that a sustained systolic blood pressure <120mmHg within 48 hours of admission was predictive of mortality and poor GOSE suggesting that an even higher systolic blood pressure target >120mmHg may be more efficacious in minimizing secondary insults (16).
A 2017 observational study of a large prehospital database established as a part of the Excellence in Prehospital Injury Care (EPIC) Traumatic Brain Injury Study determined the relationship between systolic pressure and probability of death. This study, which included 3,844 patients with moderate-to-severe TBI, described a significant linear relationship between pre-hospital systolic blood pressure and probability of mortality without offering a clear inflection point. The authors suggested that the specific cutoff value for meaningful hypotension may not exist (17).
The 2017 Brain Trauma Foundation Guidelines for the Management of Severe TBI recommend maintaining SBP at ≥ 100 mm Hg for patients 50 to 69 years old and ≥ 110 mm Hg or above for patients 15 to 49 or ≥ 70 years old to decrease mortality and improve outcomes. They also recommend using caution with mannitol to avoid dropping systolic blood pressure below 90mmHg (18).
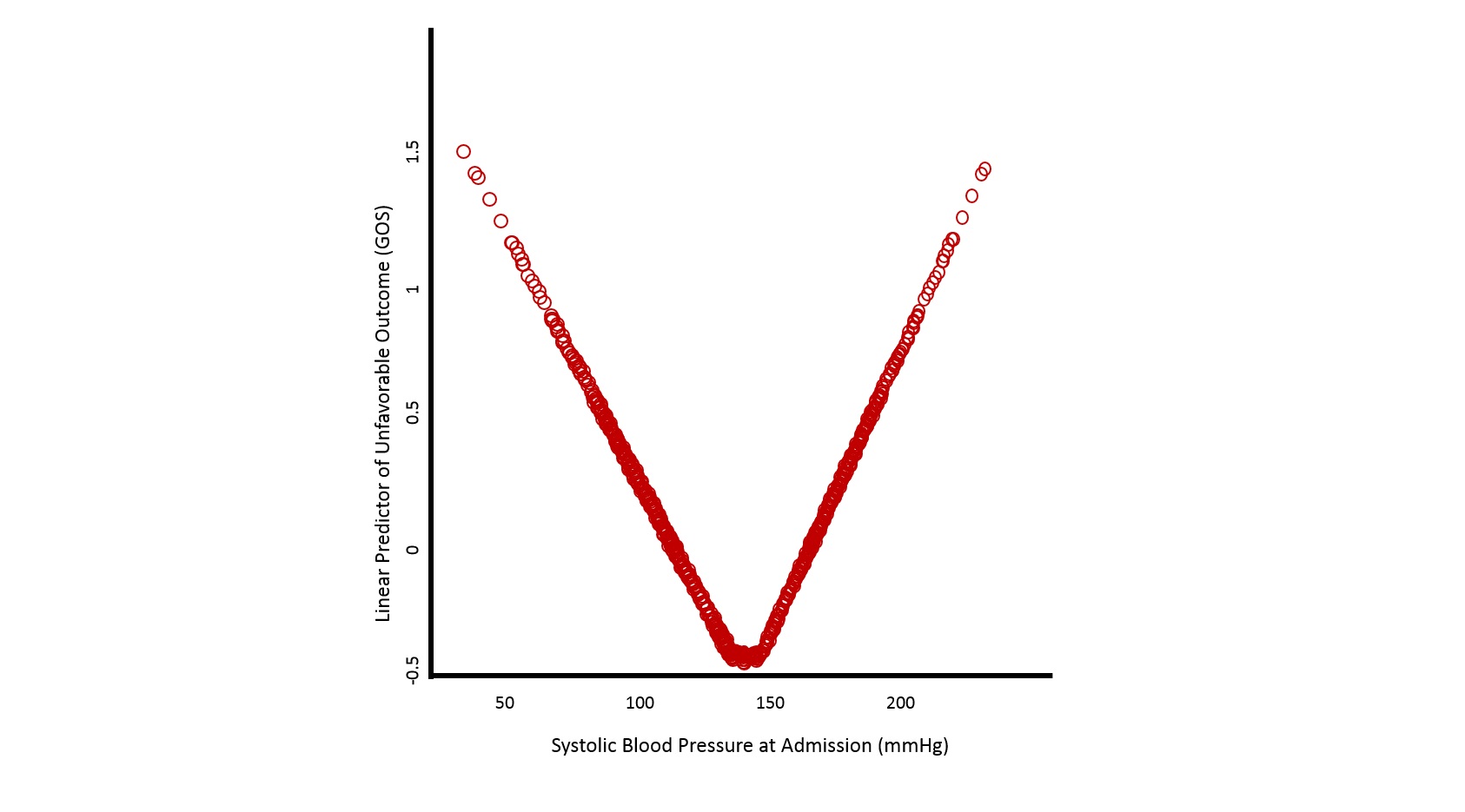
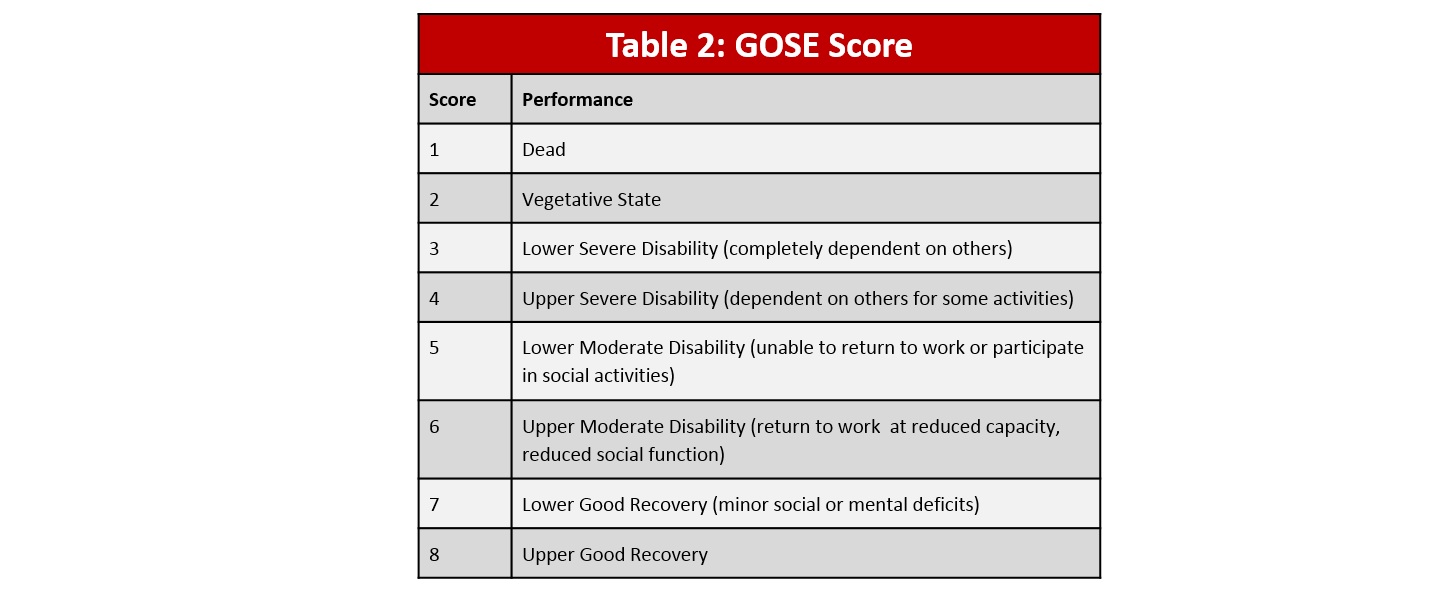
Table 2: GOSE Score; Figure 2: Relation between systolic blood pressure and Glascow Outcome Scale (GOS) Score
Hypotensive Strategies in Trauma Patient’s with Active Hemorrhage
The use of permissive hypotension was first described in the early 1900s as a strategy to minimize active bleeding. Aggressive normalization of blood pressure in the hemorrhagic shock patient may result in increased hydrostatic pressure and clot dislodgement, hypothermia, and coagulopathy worsening the lethal triad (19). “Injection of a fluid that will increase blood pressure has dangers in itself. … If the pressure is raised before the surgeon is ready to check any bleeding that might take place, blood that is sorely needed may be lost.” (20) This concept has also come to be known as damage control resuscitation (DCR) after C. William Schwab, MD, a Navy combat surgeon, who proposed this term as a comparison to the Navy term “Damage Control” which are efforts required in stabilizing an insult to a ship. Damage Control Resuscitation was included as a U.S. Department of Defense clinical practice guideline as early as 2004 and has become a standard of care in both military and civilian settings (21).
The majority of resuscitation research on permissive hypotension in patients with acute hemorrhage, or damage control resuscitation, comes from animal studies, and few trials with human subjects. Below five studies of permissive hypotension involving human subjects are reviewed:
Immediate verses Delayed Fluid Resuscitation for Hypotensive Patients with Penetrating Torso Injuries (1994)
One of the few pre-hospital resuscitation studies involving humans, a landmark study by Bickell et al. (1994), demonstrated a survival benefit in patients with penetrating torso injuries in whom fluid administration was restricted until definitive hemostasis was achieved. In this non-blinded single center prospective trial of 598 patients with penetrating torso injuries, delay of fluid resuscitation until the time of operative intervention resulted in an increase in survival (70% vs 62%, P = 0.04) and significant decrease in acute respiratory distress syndrome, mechanical ventilator time, intensive care unit length of stay, and acute renal injury (22).
Hypotensive Resuscitation during Active Hemorrhage: Impact on In-Hospital Mortality (2002)
Dutton et al. enrolled 110 subjects in a prospective randomized trial in which the control group was treated to maintain SBP > 100 mmHg whereas the treatment group was treated to maintain SBP > 70 mmHg. The subjects consisted of 79% males with 51 % penetrating trauma. Ultimately, a significant difference in SBP was observed with no difference in the injury severity score, the duration of bleeding or survival (23).
Hypotensive Resuscitation Strategy Reduces Transfusion Requirements and Severe Postoperative Coagulopathy in Trauma Patients With Hemorrhagic Shock: Preliminary Results of a Randomized Controlled Trial (2011)
Morrison et al. enrolled 90 subjects in a prospective randomized trial in which the control group was treated to maintain mean arterial blood pressures (MAP) > 65 mmHg whereas the treatment group was treated to maintain MAPs > 50 mmHg. The primary outcome was 30-day mortality. The subjects consisted of 90% males with 93.33% penetrating trauma. The patients in the treatment group (MAP > 50 mmHg) had significantly lower mortality in the early postoperative period (2.2% vs. 17.4%; p = 0.03) and a nonsignificant trend for lower mortality at 30 days (22.7% vs. 28.2%; p = 0.55) (24).
A Controlled Resuscitation Strategy is Feasible and Safe in Hypotensive Trauma Patients: Results of a Prospective Randomized Pilot Trial (2015)
Schreiber et al. enrolled 192 subjects in this prospective randomized trial in which the control group was treated with aggressive fluid resuscitation (2 L crystalloid) if the SBP was < 110 mmHg whereas the treatment group was treated with gentle fluid resuscitation (250 mL of crystalloid) for SBP < 70 mmHg. The primary outcome was in-hospital mortality. The subjects consisted of 76% males with 33.9% penetrating trauma. At 24 hours after admission there was no significant difference in mortality overall (5% vs. 15%; aOR, 0.39; 95% CI, 0.12 - 1.26) or in patients with penetrating trauma (9% vs. 9%; aOR, 1.93; 95% CI, 0.19 - 19.17). However, in patients with blunt trauma a significant difference was observed at 24 hours (3% vs. 18%; aOR 0.17; 95% CI, 0.03 - 0.92) (25).
Intraoperative Hypotensive Resuscitation for Patients Undergoing Laparotomy or Thoracotomy for Trauma: Early Termination of a Randomized Prospective Clinical Trial (2016)
Carrick et al. enrolled 168 subjects prior to early termination in a prospective randomized trial in which the control group was treated to maintain mean arterial blood pressures (MAP) > 65 mmHg whereas the treatment group was treated to maintain MAPs > 50 mmHg. Ninety percent of subjects were male, all with penetrating trauma and initial systolic blood pressure of ≤ 90 mm Hg requiring laparotomy or thoracotomy for hemorrhage control. The primary outcome was 30-day mortality with no significant difference between treatment and control groups (21.4% vs. 26.3%; p = 0.47) (26).
A 2014 Cochrane review by Kwan et al. examined the effects of time and volume of fluid resuscitation on mortality and coagulation times of trauma patients found insufficient evidence for or against the use of early or large volume fluid resuscitation in the treatment of un-controlled hemorrhage. The authors called for large, well-concealed, randomized controlled trials to establish the optimal fluid resuscitation strategy in hemorrhaging trauma patients (27).
A 2018 systematic review in The Journal of Trauma and Acute Care Surgery evaluated permissive hypotension strategies in adult trauma patients with hemorrhagic shock including five randomized trials and 1,158 patients. In the permissive hypotension (intervention) arms, target SBP and MAPs were 70 -90 mm Hg and ≥ 50 mm Hg, respectively. Within these studies the control arm target SBP and MAPs were 100 - 110 mmHg and ≥ 65 mmHg, respectfully. Two studies evaluated only patients with penetrating injury while the remaining three additionally included both penetrating and blunt injuries. Four trials suggested a survival benefit for 30-day or in-hospital mortality with hypotensive resuscitation, although three studies were insufficiently powered. Studies were of poor to moderate quality due to poor protocol reporting and lack of blinding. The pooled odds ratio was 0.70 (95% confidence interval, 0.53–0.92), suggesting a survival benefit for permissive hypotension. Additionally, the patients who were treated with hypotensive strategies received fewer blood products and had less estimated blood loss. However, ultimately the authors concluded that more adequately powered trials are needed to make definitive conclusions (28).
What is the optimal Mean Arterial Pressure for Patients with Uncontrolled Hemorrhage and TBI?
The challenge for clinicians involved in the management of trauma patients is to balance the need for cerebral perfusion against increases in systemic blood pressure that may worsen uncontrolled hemorrhage. In patients with both TBI and active systemic hemorrhage, the optimal MAP target is not clear, and there is extremely limited data on this topic. Most of the studies investigating trauma resuscitation target endpoints exclude patients with traumatic brain injury.
Another challenge for emergency medical providers is lack of concrete data on the presence or absence of intracranial hypertension. Providers in this setting must weigh the risks and benefits of certain blood pressure targets without having concrete data upon which to base their therapies. A few societies have published guidelines on optimal blood pressure targets in hemorrhagic shock and traumatic brain injury, which are summarized in Table 4.
Summary
There is little data to help us in optimal blood pressure targets in patients with both TBI and systemic trauma with hemorrhagic shock, largely because these patients are excluded from the majority of studies. Given the significant evidence that permissive hypotension would be associated with worse TBI related outcomes, we would not recommend prehospital permissive hypotension in the subset of patients with blunt, severe TBI.
In the resuscitation of these patients, hypertonic saline offers great promise as a means to resuscitate patients in hemorrhagic shock, while also minimizing intracranial pressure. Mannitol is a less than ideal alternative until more definitive data is obtained on injuries, and the patient is hemodynamically stabilized.
Future directions on this topic should include high-quality, high-powered studies investigating the hypotensive strategies to include patients with severe traumatic brain injury with GCS 8 or lower.
Finally, a good neurologic exam to more sensitively assess for focal neurologic deficits and signs of intracranial hypertension can assist pre-hospital providers to better determine the likelihood of the presence or absence of traumatic brain injury.
Authors: Kelly A. Rath, ACNP; Adam Gottula, MD
Kelly Rath is a nurse practitioner in the Neuroscience ICU. Adam Gottula, MD is a third year resident in the Department of Emergency Medicine.
Faculty Editor: Natalie Kreitzer, MD, MS
Natalie Kreitzer, MD, MS is an attending in the Department of Emergency Medicine and the Neuroscience ICU.
References:
Menon DK, et al. Position statement: definition of traumatic brain injury. Arch Phys Med Rehabil. 2010;91(11):1637–40.
Taylor CA, Bell JM, Breiding MJ, Xu L: Traumatic Brain Injury–Related Emergency Department Visits, Hospitalizations, and Deaths — United States, 2007 and 2013. MMWR Surveill Summ 66:1-16, 2017
Korley FK, Kelen GD, Jones CM, Diaz-Arrastia R: Emergency Department Evaluation of Traumatic Brain Injury in the United States, 2009-2010. J Head Trauma Rehabil 31:379-387, 2016
Faul MXL, Wald MM, Coronado VG. Traumatic brain injury in the United States: emergency department visist, hospitalizations, and deaths. Atlanta: Centers for Disease Control and Prevention, National Center for Injury Prevention and Control; 2010.
Tien HC, Spender F, Tremblay LN, Rizoli SB, Brenneman FD. Preventable deaths from hemorrhage at a level I Canadian trauma center. J Trauma. 2007; 62:142-146.
Mokri B. The monro-kellie hypothesis – applications in CSF volume depletion. Neurology. 2001;56:1746-48.
Czosnyka M, Smielewski P, Piechnik S, Steiner LA, Pickard JD. Cerebral autoregulation following head injury. J Neurosurg. 2001;95:756–63.
Baron JC. Perfusion thresholds in human cerebral ischemia: historical perspective and therapeutic implications. Cerebrovasc Disease; 2001: 11: 2-8 (supp).
Leung LY, Wei G, Shear DA, Tortella FC. The acute effects of hemorrhagic shock on cerebral blood flow, brain tissue oxygen tension, and spreading de- polarization following penetrating ballistic-like brain injury. J Neurotrauma. 2013;30:1288–1298.
Oswal, A, Toma AK. Intracranial pressure and cerebral haemodynamics. Anaes Intens Care Med, 2017; 18(5):259-63.
Chesnut RM, Marshall LF, Blunt BA, Baldwin N, Eisenberg HN, Jane JA, Marmarou A, Foulkes MA. The role of secondary brain injury in determining outcome from severe head injury. J Trauma. 1993: 34: 216-222.
Wald SL, Shackford SR, Fenwick J. The effects of secondary insults on mortality and long-term disability after severe head injury in a rural region without a trauma system. J Truama. 1993: 34: 377-382.
McHugh, GS, Engel DC, Butcher I, Steyerberg EW, Lu J, Mushkudiani N, Hernandez AV, Marmarou A, Maas AJR, Murray GD. Prognostic value of secondary insults in traumatic brain injury: results from the IMPACT study. J Neurotrauma. 2007;24(2):287-93.
Berry C, Ley EJ, Bukur M, Malinoski D, Margulies DR, Mirocha J, Salim A. Redefining hypotension in traumatic brain injury. Injury. 2012; 43: 1833-37.
Butcher I, Maas AIR, Lu J, Marmarou A, Murray GD, Mushkudiani NA, McHugh GS, Steyerberg EW. Prognostic value of admission blood pressure in traumatic brain injury: results from the IMPACT study. J Neurotrauma. 2007; 24(2):294-302.
Maas AIR, Murray GD, Lingsma HF, Butcher I, McHugh GS, Weir J, Lu J, Steyerbert EW. Advancing care for traumatic brain injury: findings from the IMPACT studies and perspectives on future research. The Lancet. 2013; 12:1200-10.
Brenner M, Stein DM, Hu PF, Aarabi B, Sheth K, Scalea TM. Traditional systolic blood pressure targets underestimate hypotension-induced secondary brain injury. J Trauma. 2012; 72(5):1135-39.
Spaite DW, et al. Mortality and prehospital blood pressure in patients with major traumatic brain injury: implications for the hypotension threshold. JAMA Surg. 2016;152:360–8.
Carney N, Totten AM, O’Reilly C, Ullman JS, Hawryluk GWJ, Bell MJ, Bratton20 SL, Chesnut R, Harris OA, Kissoon N, Rubiano AM, Shutter L, Tasker RC, Vavilala MS, Wilberger J, Wright DW, Ghajar J. Guidelines for the management of severe traumatic brain injury, fourth edition. Neurosurgery. 2017;80(1):6-15.
Wiles MD. Blood pressure in trauma resuscitation: ‘pop the clot’ vs. ‘drain the brain’ - editorial. Anaesthesia. 2017; 72:1435–1455.
Dauer E, Goldberg A. What’s new in trauma resuscitation? Advances in Surgery. 2019: in press.
Sklar R, Han K, International Trauma Life Support: Damage Control Resuscitation and Management of Severe Hemorrhage/Shock in the Preshopital Setting. 2019
Bickell WH, Wall MJ, Pepe PE, Martin RR, Ginger VF, Allen MK, Mattox KL. Immediate versus delayed fluid resuscitation for hypotensive patients with penetrating torso injuries. NEJM. 1994;331(17):1105-09.
Dutton RP, Mackenzie CF, Scalea TM. Hypotensive resuscitation during active hemorrhage: impact on in-hospital mortality. J Trauma. 2002 Jun;52(6):1141-6.
Morrison CA, Morrison CA1, MM, Norman MA, Scott BG, Welsh FJ, Tsai P, Liscum KR, Wall MJ Jr, Mattox KL. Hypotensive resuscitation strategy reduces transfusion requirements and severe postoperative coagulopathy in trauma patients with hemorrhagic shock: preliminary results of a randomized controlled trial. J Trauma. 2011; 70(3): 652-63.
Schreiber MA, Meier EN, Tisherman SA, Kerby JD, Newgard CD, Brasel K, Egan D, Witham W, Williams C, Daya M, et al. A controlled resuscitation strategy is feasible and safe in hypotensive trauma patients: results of a prospective randomized pilot trial. J Trauma Acute Care Surg. 2015;78(4):687–695
Carrick MM, Morrison CA, Tapia NM, Leonard J et al. Intraoperative hypotensive resuscitation for patients undergoing laparotomy or thoracotomy for trauma: Early termination of a randomized prospective clinical trial. J Trauma Acute Care Surg. 2016; 80(6):886-96.
Kwan I, Bunn F, Chinnock P, Roberts I. Timing and volume of fluid administration for patients with bleeding (review). Cochrane Database of Systematic Reviews. 2014; 3: 1-16.
Tran A, Yates J, Lau A, Lampron J, Matar M. Permissive hypotension vs conventional resuscitation strategies in adult trauma patients with hemorrhagic shock: A systematic review and meta-analysis of randomized controlled trials. J Trauma Acute Care Surg. 2018; 84(5):802-808.
Roissant et al. The European guideline on management of major bleeding and coagulopathy following trauma: fourth edition. Critical Care. 2016; 20(100):1-55.
NICE guideline Published: 17 February 2016. nice.org.uk/guidance/ng39
Garver R, Mangat HS. Emergency Neurologic Life Support: Severe Traumatic Brain Injury. Neurocrit Care. 2017.
Advanced Trauma Life Support : 10th Edition (2018). American College of Surgeons




