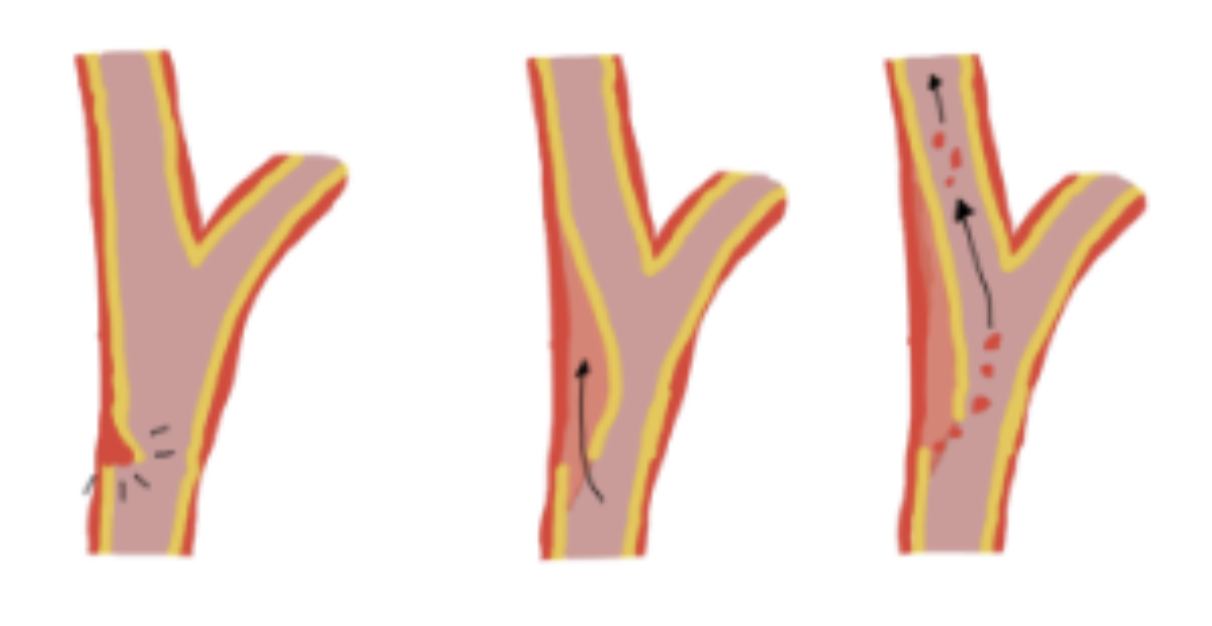Blunt Cerebrovascular Injuries
/Blunt cerebrovascular injuries can often be subtle and difficult to recognize and diagnose. They are associated with significant morbidity and mortality. Therefore, early recognition, diagnosis and treatment of these injuries is vital. In this post we will explore blunt cerebrovascular injuries, the nuances associated with them and how to properly manage these patients.
Overview
Blunt cerebrovascular injury (BCVI) refers to non‑penetrating trauma causing injury to the carotid or vertebral arteries, with risk of stroke from thrombosis, dissection, or pseudoaneurysm. BCVIs are often clinically silent early, but with high morbidity when missed. Most commonly, these injuries occur from high energy mechanisms
Prior to availability of screening tests, blunt carotid injury was associated with mortality rates ranging from 23 to 28 percent, with 48 to 58 percent of survivors suffering from permanent severe neurologic deficits. With the increased development of diagnostic screening based on certain criteria, the reported incidence of BCVIs in patients with blunt trauma has increased approximately 1-3%.
Basic Anatomy
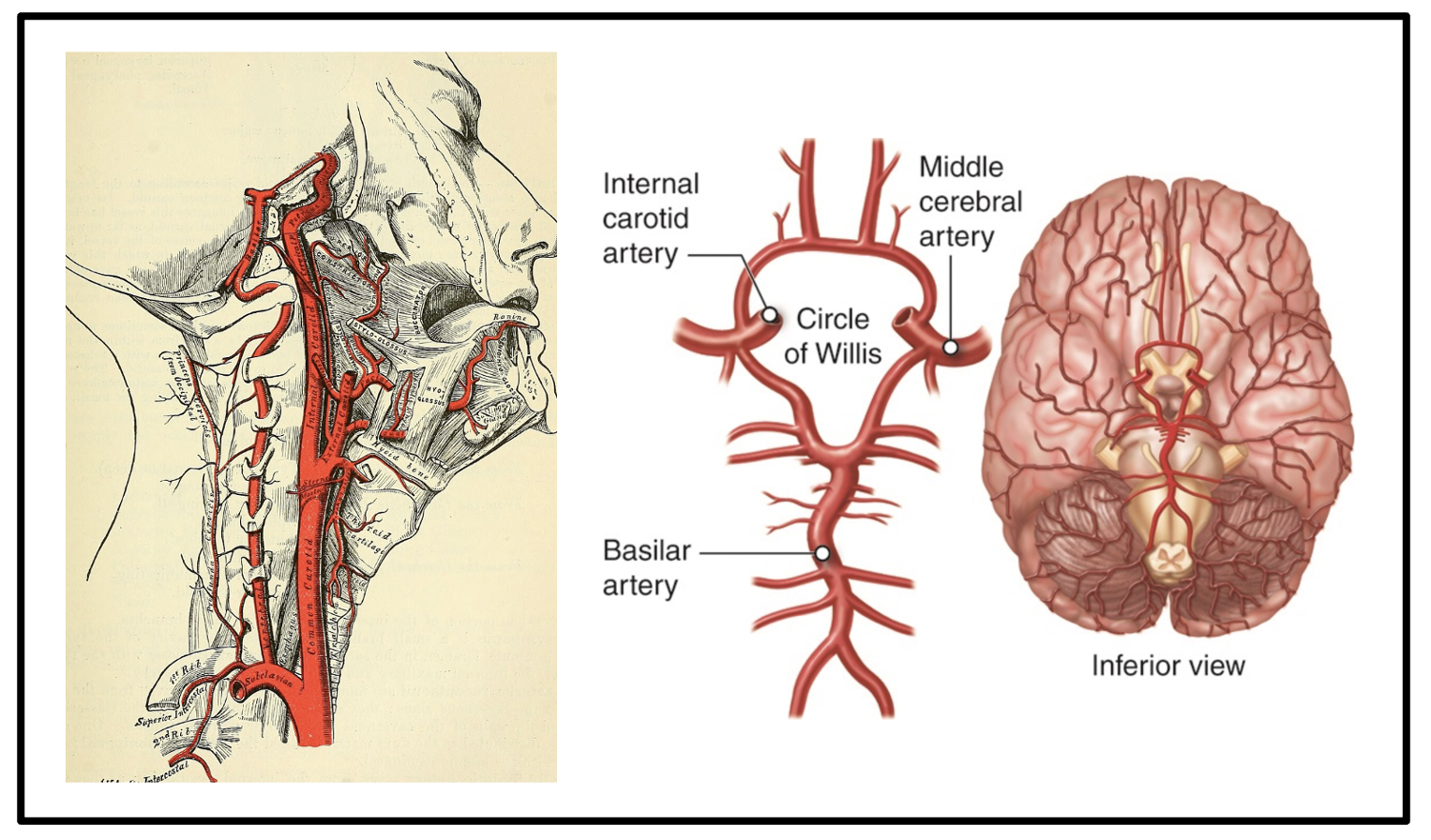
The vascular supply to the brain is divided into the anterior and posterior circulations
The anterior circulation is supplied by the carotid arteries.
The posterior circulation is supplied by the vertebral arteries, which take a course parallel to the spinal column through the transverse foramina.
The circle of Willis connects the anterior and posterior circulations, but is completely intact and symmetric in only approximately 20 percent of individuals. This variability is important to consider when BCVI is suspected in trauma patients causing a wide variety of clinical presentations.
Risk Factors and Mechanisms of Action of Injury
There are a variety of mechanisms that can lead to BCVIs, described below with matching pictures showing each mechanism.
(A) Intimal tearing caused by cervical hyperextention stretching the internal carotid artery across the processes of the first through third cervical vertebrae.
(B) Intimal disruption caused by cervical hyperflexion directly compressing the internal carotid artery between the mandible and upper cervical vertebrae.
(C) Falling with a hard object in the mouth (eg, toothbrush) can lead to injury of the internal carotid artery behind the soft palate at the angle of the jaw.
(D) Trauma to the anterior neck can cause a direct blow injury to the carotid artery.
(E) A basilar skull fracture can cause injury to the carotid or vertebral arteries as they enter the skull base.
These types of injuries leading to BCVIs are most commonly caused by the following situations:
High‑velocity MVCs (account for over half of BCVIs)
Ground level falls (account for more than ⅔ of BCVIs in patients over 65 years old)
Direct blunt neck trauma
Basilar skull fractures involving carotid canal
LeFort II/III facial fractures
Cervical spine subluxation/rotation injuries
Of note, cervical spine fractures have the strongest association with associated BCVI, according to a meta-analysis, unrelated to the degree of energy transfer or mechanism. This is due to the proximity of the vertebral artery to the cervical spine as it runs parallel to the spinal column through the transverse foramina.
Regardless of the underlying mechanism of injury, the pathologic insult is usually an intimal tear. The exposed subendothelial collagen promotes platelet aggregation and thrombus formation, which may occlude the vessel altogether or embolize it to the cerebral circulation. BCVI is often associated with high-energy transfer mechanisms that can cause severe head, facial, spine, chest, and abdominal injuries. Therefore it is important to evaluate for other injuries in patients with BCVI.
Clinical Presentation
General Timeline and Symptoms
Most patients with a BCVI are asymptomatic from a neurologic standpoint on initial presentation.
There is typically a latent period between the initial injury and the appearance of clinical manifestations.
In many studies between 25 and 50% of patients did not develop neurologic symptoms of BCVI until more than 12 hours after the traumatic event.
The symptoms seen on neurologic examination depend on on the artery involved as well as the presence of absence of adequate collateral circulation
When patients do experience symptoms, they often experience neck, face, ear, periorbital pain (appx 60%) with carotid or vertebral artery dissection in the neck
Approximately 10 to 15 percent of symptomatic patients have lateralizing neurologic symptoms to the contralateral side
No single neurologic finding allows a precise diagnosis, but the constellation of findings may identify the involved artery
Diagnostics
Neurovascular imaging is indicated for patients with any clinical symptom or sign suggestive of BCVI and should be considered in patients with unexplained neurologic signs or symptoms.
The modified Memphis criteria are a set of screening criteria for blunt cerebrovascular injury (BCVI) in trauma. The presence of one or more of these criteria makes it necessary to perform a complementary CTA or DSA study to exclude a BCVI.
High-Risk Symptoms:
Arterial hemorrhage from neck/nose/mouth
Expanding neck hematoma
Focal neurologic deficit not explained by CT head
Stroke on initial CT head in trauma context
Ptosis, miosis, anhidrosis (Horner Syndrome)
Cervical bruit or thrill (in patients <50)
High-Risk Injury Patterns:
C1–C3 fractures
Cervical spine subluxation or rotation injury
Fractures through the foramen transversarium
LeFort II/III facial fractures
Basilar skull fracture involving carotid canal
Seatbelt abrasion with significant swelling or pain
Frontal skull fractures extending into the sphenoid
The imaging modality of choice is CTA of the neck to evaluate for vertebral and carotid artery dissection. Although CT angiography may lack sensitivity to completely rule out blunt cerebrovascular injury, it remains the best study for emergency department patients who have a high pretest probability of injury (ie, appropriate risk factors). MRA is available if unable to obtain CTA.
Grading of Injury Patterns
A grading scale for blunt carotid artery injury standardizes clinical communication and helps guide therapy. This scale titled “Denver Injury Grades” was developed based on conventional arteriography findings and has been validated for computed tomographic (CT) angiography use only.
Grade I – Intimal irregularity or dissection with <25 percent luminal narrowing.
Grade II – Dissection or intramural hematomas with ≥25 percent luminal narrowing, intraluminal clot, or a visible intimal flap.
Grade III – Pseudoaneurysm or hemodynamically insignificant arteriovenous fistula.
Grade IV – Complete occlusion.
Grade V – Transection with active extravasation (hemorrhage) or hemodynamically significant arteriovenous fistula. These injuries are often lethal. Transection injuries mandate immediate attempts to control bleeding.
Management
The nature of symptoms, injury location, and specific type (grade) of injury may indicate the need for other interventions in addition to antithrombotic therapy. The effectiveness of antithrombotic therapy has obviated the need for open surgical or endovascular interventions for most patients, particularly those who are asymptomatic.
First‑line options:
Aspirin (81–325 mg daily for grades I-III), these patients can often be sent home after neurosurgical consultation
Heparin infusion (low-dose, no bolus) targeting PTT 40–50 (for grade IV), these patients require admission after neurosurgical consultation
Surgical revision usually required for grade V injuries, especially when bleeding is unable to be controlled
Evidence: Both anticoagulation and antiplatelets reduce stroke risk; selection depends on bleeding risk.
Grade‑based summary:
Grade I–II: Antithrombotic therapy
Grade III: Antithrombotic therapy; consider endovascular repair for enlarging pseudoaneurysm
Grade IV: Antithrombotic therapy, stroke prevention
Grade V: Usually requires urgent operative or endovascular intervention
If major bleeding/trauma contraindicates therapy:
Involve trauma + neurointerventional early
Start antiplatelet therapy as soon as safe
FOLLOW‑UP IMAGING
Typical schedule:
Repeat CTA at 7 days to assess progression or healing
Repeat at 3 months to guide duration of therapy
Many Grade I–II injuries heal completely
Key Points
Consider BCVI if you see:
Unexplained lateralizing neurologic signs
Stroke pattern that doesn't match head CT findings
High-energy mechanism with neck or facial fractures
Severe neck pain after high‑speed MVC even with normal CT C‑spine
Common Pitfalls:
Failing to scan because patient is asymptomatic
Missing subtle fracture patterns
Delaying antithrombotic therapy due to "fear of bleeding" without evaluating actual risk
Not repeating imaging to track progression
Post by : Maxwell king, MD
Dr. King is a PGY-1 in Emergency Medicine at the University of Cincinnati
Editing by : anita Goel, MD
Dr. Anita Goel is an APD in Emergency Medicine at the University of Cincinnati and Co-editor of Tamingthesru.com
References
Biffl WL, Moore EE, Ryu RK, et al. The unrecognized epidemic of blunt carotid arterial injuries: early diagnosis improves neurologic outcome. Ann Surg 1998; 228:462.
Kelts G, Maturo S, Couch ME, Schmalbach CE. Blunt cerebrovascular injury following craniomaxillofacial fractures: A systematic review. Laryngoscope 2017; 127:79.
Shahan CP, Gray RI, Croce MA, Fabian TC. Impact of circle of Willis anatomy in traumatic blunt cerebrovascular injury-related stroke. Trauma Surg Acute Care Open 2017; 2:e000086.
Cogbill TH, Moore EE, Meissner M, et al. The spectrum of blunt injury to the carotid artery: a multicenter perspective. J Trauma 1994; 37:473.
Biffl WL, Ray CE Jr, Moore EE, et al. Treatment-related outcomes from blunt cerebrovascular injuries: importance of routine follow-up arteriography. Ann Surg 2002; 235:699.
Edwards NM, Fabian TC, Claridge JA, et al. Antithrombotic therapy and endovascular stents are effective treatment for blunt carotid injuries: results from longterm followup. J Am Coll Surg 2007; 204:1007.
Anto VP, Brown JB, Peitzman AB, et al. Blunt cerebrovascular injury in elderly fall patients: are we screening enough? World J Emerg Surg 2018; 13:30.
Cook A, Osler T, Gaudet M, et al. Blunt cerebrovascular injury is poorly predicted by modeling with other injuries: analysis of NTDB data. J Trauma 2011; 71:114.
Franz RW, Willette PA, Wood MJ, et al. A systematic review and meta-analysis of diagnostic screening criteria for blunt cerebrovascular injuries. J Am Coll Surg 2012; 214:313.
Lebl DR, Bono CM, Velmahos G, et al. Vertebral artery injury associated with blunt cervical spine trauma: a multivariate regression analysis. Spine (Phila Pa 1976) 2013; 38:1352.
Cothren CC, Biffl WL, Moore EE, et al. Treatment for blunt cerebrovascular injuries: equivalence of anticoagulation and antiplatelet agents. Arch Surg 2009; 144:685.
Biffl WL, Moore EE, Offner PJ, et al. Blunt carotid arterial injuries: implications of a new grading scale. J Trauma 1999; 47:845.
Burlew CC, Sumislawski JJ, Behnfield CD, et al. Time to stroke: A Western Trauma Association multicenter study of blunt cerebrovascular injuries. J Trauma Acute Care Surg 2018; 85:858.
Biffl WL, Moore EE. Computed tomographic angiography for blunt cerebrovascular injuries: don't throw out the baby with the bathwater. Ann Surg 2011; 253:451.
Biffl WL, Moore EE, Offner PJ, et al. Blunt carotid arterial injuries: implications of a new grading scale. J Trauma 1999; 47:845.
Foreman PM, Griessenauer CJ, Kicielinski KP, et al. Reliability assessment of the Biffl Scale for blunt traumatic cerebrovascular injury as detected on computer tomography angiography. J Neurosurg 2017; 127:32.